2a Review How Does the Immune System Identify a Pathogen?
Infectious diseases are a leading crusade of morbidity and bloodshed worldwide and are a major claiming for the biomedical sciences. Improved sanitary conditions, clean h2o supplies and vector control are by far the virtually effective measures to reduce the incidence of infectious disease. Still, the development of vaccines and therapeutics is also of import, and this requires an understanding of the host immune system. Recently, much progress has been made towards discovering the mechanisms of microbial pathogenesis and host–microbe symbiosis. And knowledge most the immune system has besides been steadily increasing. Yet many challenges remain, maybe the most daunting being constructive vaccine evolution. Indeed, it is non known how to elicit protective immunity confronting most pathogens in a safe and practical fashion. To accomplish this and other goals, such equally the rubber and efficient occludent of autoimmune and allergic allowed responses, further developments in bones research are clearly required.
Here, I provide a general overview of the immune system every bit it relates to defence against microorganisms, with an emphasis on recent findings.
Host–microbe interactions
All metazoan hosts exist in close association with microbial communities that colonize them. The 'rules of engagement' of host–microbe interactions are incompletely understood, and defining these is conspicuously of import for agreement the evolution and functioning of the immune system.
The host every bit a set of niches colonized past microorganisms
Mammalian hosts provide a number of niches that can be colonized by microorganisms, including the pare, intestine, upper and lower respiratory tract, urogenital tract and internal organs. Some of these niches (for case, the colon and the skin) are colonized constitutively by an endogenous microbiota. Other niches (for instance, the internal organs and the lower respiratory tract) are normally kept sterile (in an immunocompetent host). The upshot of microbial colonization on host fitness depends on the microbial adaptation strategy. These effects can be positive, equally is the case for the many intestinal bacteria that provide a range of benefits to the host (come across pages 804 and 811). In other cases, microbial colonization can be detrimental to the host, and these colonizing leaner are referred to as pathogens. Such negative effects can depend on the status of the host's immune organisation: for example, sure pathogens, known as opportunistic pathogens, affect only immunocompromised individuals.
Virulence factors
The adaptation of bacteria to particular host niches depends on the activeness of various accommodation factors; for pathogens, these are known every bit virulence factors. Accommodation factors are often encoded on mobile genetic elements (for example, plasmids and genomic islands) that tin exist transmitted within and between bacterial speciesone (see page 835), although there are important exceptions (for instance, in Mycobacterium spp.)2. The role of virulence factors is to enable adaptation to the specific environments in the host niches and to promote transmission to another host. In this way, some common themes of virulence-factor activity (and therefore pathogenicity) can be identified3. Depending on the niche that they colonize, bacterial pathogens accept virulence factors that allow a range of activities: penetration of surface epithelia, attachment to cell surfaces and/or the extracellular matrix, invasion of intracellular compartments, acquisition of iron, evasion of host-defense mechanisms and manual to another host. Unlike strategies of pathogenic microbial adaptation are associated with varying degrees of damage to the tissues of the host. Regardless of the caste of virulence, at least some symptoms of communicable diseases are side-effects of microbial adaptation to host niches.
Recognition of microorganisms by the immune system
The detrimental effects of microbial infections led to the evolution of a variety of host-defence force mechanisms. In jawed vertebrates, there are ii types of defence: innate and adaptive (also known as acquired). The primary distinction between these is the receptor types used to recognize pathogens. Innate immune recognition is mediated by pattern-recognition receptors (PRRs), which are germline encoded, and each receptor has broad specificities for conserved and invariant features of microorganisms4. By dissimilarity, adaptive immune recognition is mediated by antigen receptors: the genes encoding these receptors are assembled from factor segments in the germ line, and somatic recombination of these segments enables the generation of a diverse repertoire of receptors with random but narrow specificities5. Antigen receptors are clonally distributed on T and B lymphocytes, which allows clonal selection of pathogen-specific receptors and is the ground for immunological memory. (That is, each lymphocyte expresses antigen receptors of a single specificity, so merely specific populations of lymphocytes are selected to aggrandize in response to a pathogen.) Therefore, the innate allowed arrangement and the adaptive immune system bargain with the molecular diversity of pathogens in fundamentally unlike means.
Innate immune system
Innate immune recognition (likewise known as design recognition) is based on the detection of molecular structures that are unique to microorganisms4. Design recognition is unusual in that each host receptor (PRR) has a wide specificity and tin can potentially bind to a large number of molecules that take a common structural motif or pattern. The targets of PRRs are sometimes referred to as pathogen-associated molecular patterns (PAMPs), although they are nowadays on both pathogenic and non-pathogenic microorganisms. PAMPs are well suited to innate immune recognition for iii principal reasons. First, they are invariant amid microorganisms of a given class. Second, they are products of pathways that are unique to microorganisms, allowing bigotry between self and non-self molecules. Third, they have essential roles in microbial physiology, limiting the ability of the microorganisms to evade innate immune recognition through adaptive evolution of these molecules. Bacterial PAMPs are often components of the cell wall, such equally lipopolysaccharide, peptidoglycan, lipoteichoic acids and cell-wall lipoproteins. An important fungal PAMP is β-glucan, which is a component of fungal prison cell walls. The detection of these structures past the innate immune organisation can betoken the presence of microorganisms. The recognition of viruses also partly follows this principle. However, because all viral components are synthesized inside host cells, the main targets of innate immune recognition in this example are viral nucleic acids. Discrimination between self (host) and viral nucleic acids occurs on the basis of specific chemical modifications and structural features that are unique to viral RNA and Dna, as well as on the cellular compartments where viral (just non host-derived) nucleic acids are normally establish (discussed later). Still, this discrimination is non perfect and tin can neglect under sure conditions, which can result in the development of autoimmune diseasessix.
An important aspect of pattern recognition is that PRRs themselves exercise not distinguish betwixt pathogenic microorganisms and symbiotic (not-pathogenic) microorganisms, because the ligands of the receptors are not unique to pathogens. Yet, despite humans beingness colonized by trillions of symbiotic bacteria, homeostasis is somehow maintained under normal conditions. Furthermore, innate immune recognition of symbiotic microorganisms has an of import role in maintaining intestinal homeostasis7. And dysregulation of these interactions can lead to the development of inflammatory bowel disease and other disorders.
PRRs and their functions
There are several functionally distinct classes of PRR (Table 1). The best characterized class is Toll-like receptors (TLRs). TLRs are transmembrane receptors that recognize viral nucleic acids and several bacterial products, including lipopolysaccharide and lipoteichoic acids (run into ref. 8 for a review). The full range of TLR functions in antimicrobial defence force has non yet been determined, simply TLRs are known to elicit inflammatory and antimicrobial responses after activation by their microbial ligands.
In terms of the inflammatory response, TLRs activate tissue-resident macrophages to produce pro-inflammatory cytokines, including neoplasm-necrosis factor (TNF), interleukin-1β (IL-1β) and IL-half dozen, which coordinate local and systemic inflammatory responses. TNF and IL-1β, in turn, activate the local endothelium to induce vasodilation and increase the permeability of the claret vessel, assuasive serum proteins and leukocytes to be recruited to the site of infection. In addition, an increase in the amount of tissue factor (also known as coagulation gene Iii) on the endothelium leads to a local coagulation cascade that helps to prevent microbial dissemination through the claret. Furthermore, IL-1β, together with IL-6, activates hepatocytes to produce acute-phase proteins, including collectins and pentraxins. These proteins, in turn, activate complement and opsonize pathogens for phagocytosis by macrophages and neutrophils. In this style, TLRs indirectly arm-twist an antimicrobial response.
TLRs likewise directly trigger such a response, by inducing macrophages to produce antimicrobial proteins and peptides. In mouse macrophages, the activation of TLRs results in transcription of the gene encoding inducible nitric-oxide synthase (iNOS; too known equally NOS2), which has an important role in antimicrobial defenceix. Interestingly, iNOS is not produced in response to the activation of TLRs on human being macrophages10. Instead, homo keratinocytes synthesize vitamin D, which is crucial for antimicrobial activeness, partly because of vitamin-D-receptor-dependent induction of the cistron encoding the antimicrobial peptide LL37 (too known as Campsite)11. Sunlight (a source of UVB radiations) is necessary for vitamin D synthesis, so the difference in vitamin D requirement for antimicrobial defence might reverberate the nocturnal and diurnal lifestyles of mice and humans, respectively.
Some other well-characterized PRR is dectin 1, a transmembrane receptor that binds to β-glucan12 and is nowadays on dendritic cells and macrophages. Dectin ane is a fellow member of a large family of C-blazon lectins, many of which are nowadays on these aforementioned prison cell types but have unknown functions13. Dectin 1 contains an singular immunoreceptor tyrosine-based activation motif (ITAM) that engages the protein tyrosine kinase SYK, thereby activating a signalling pathway that involves CARD9, Bcl-10 and MALT1 (ref. 14). This PRR has an important role in antifungal defence force15,16, beingness involved in the phagocytosis of fungal pathogens, the induction of an antimicrobial response (such equally activation of NADPH oxidase) and the production of cytokines17.
In addition to transmembrane receptors on the cell surface and in endosomal compartments, at that place are intracellular (cytosolic) receptors that role in the design recognition of bacterial and viral pathogens. These include NLRs and the intracellular sensors of viral nucleic acids RIG-I (retinoic-acid-inducible gene I; also known as DDX58), MDA5 (melanoma differentiation-associated gene 5; also known as IFIH1) and DAI (DNA-dependent activator of interferon-regulatory factors; also known as ZBP1). NLRs are a big family of nigh 20 intracellular proteins with a common protein-domain organization merely diverse functions18,19,20,21. All NLRs incorporate a nucleotide-binding oligomerization domain (NOD) followed by a leucine-rich-echo domain at the carboxy terminus. At the amino terminus, NLRs have i of 3 domains and are thereby categorized into 3 subfamilies: a caspase-recruitment domain (CARD), present in proteins in the NOD subfamily; a pyrin domain, in the NALP subfamily; or a BIR domain (baculoviral inhibitor-of-apoptosis-protein echo-containing domain), in the NAIP subfamily18,xix,20,21. The North-last domains appoint distinct signalling pathways, which define the functional properties of the family members.
The proteins of the NOD subfamily — NOD1 and NOD2 — are both involved in sensing bacterial peptidoglycans, although they recognize structurally distinct peptidoglycan fragments18. The sensing of peptidoglycan by NOD1 or NOD2 triggers the production of pro-inflammatory cytokines and chemokines and the recruitment of neutrophils to the site of infection19. In addition, these NOD proteins contribute to the initiation of the adaptive allowed response22,23, and mutations in NOD2 have been implicated in the pathogenesis of Crohn'southward disease24. NOD2 is likewise crucial for the production of antimicrobial peptides known every bit defensins past Paneth cells (which are present in the pocket-sized intestine), and NOD proteins tin presumably activate antimicrobial responses in other cell types23.
The NALP subfamily of NLRs has 14 members, and at least some of these are involved in the consecration of the inflammatory response mediated past the IL-ane family of cytokines, which includes IL-1β, IL-18 and IL-33 (ref. twenty). These cytokines are synthesized as inactive precursors that need to be cleaved by the pro-inflammatory caspases: that is, caspase ane, caspase 4 and caspase five in humans, and caspase 1, caspase eleven and caspase 12 in mice. These caspases are activated in a multisubunit complex chosen the inflammasome25. In that location are several types of inflammasome, categorized co-ordinate to their composition and the interest of a particular NALP or NAIP. The private inflammasomes are activated in response to a multifariousness of bacterial infections, past mechanisms that accept been poorly divers20. Why IL-ane-family members are activated by such an elaborate mechanism is puzzling. Unlike other pro-inflammatory cytokines, IL-1β production is regulated by two singled-out signals: TLR-induced transcription and inflammasome-mediated processing of the precursor protein. Information technology is possible that, in addition to IL-1-family members, the inflammasomes process antimicrobial peptides or proteins that take not yet been characterized. NALPs might also contribute to antimicrobial defence by inducing the apoptosis of infected cellstwenty. Whether they can also directly induce the expression of antimicrobial genes is unknown.
Intracellular recognition of viral infections is mediated by two types of viral nucleic-acid sensor. Viral RNA in the cytosol is detected by the RNA-helicase-family proteins RIG-I and MDA5 (ref. 26), whereas viral Dna is detected by the recently identified poly peptide DAI27. RIG-I and MDA5 recognize different types of viral RNA: single-stranded RNA containing v′ triphosphate and double-stranded RNA, respectively28,29,30,31. These structural features are absent-minded from cellular (host) RNAs, which contain either short hairpin structures, in the example of transfer RNAs and ribosomal RNAs, or a 5′-cap structure, in the instance of messenger RNA. These structural differences allow bigotry betwixt viral and cocky RNAs. Activation of RIG-I or MDA5 results in the production of type I interferons (IFNs; IFN-α and IFN-β) and thereby the induction of antiviral immunityxxx. Interestingly, a crucial adaptor involved in RIG-I and MDA5 signalling is associated with the mitochondrial membrane32, merely the reason for this is unclear at present. The details of how viral Deoxyribonucleic acid is recognized in the cytosol, and the signalling pathways induced by the engagement of DAI, are not yet known. It is, however, articulate that the RNA-sensing pathway and the DNA-sensing pathway converge on the protein kinase TBK1 (TANK-binding kinase i) and the transcription factor IFN-regulatory factor 3 (refs 33,34,35). Type I IFNs are therefore elicited by the date of either type of sensor. This results in antiviral immune responses in both cases, through inducing the expression of numerous IFN-inducible genes, the products of which have a broad range of antiviral activities36.
Adaptive immune arrangement
Adaptive immune recognition is mediated by two types of antigen receptor: T-prison cell receptors and B-cell receptors. The genes encoding antigen receptors are assembled from variable and constant fragments through recombination-activating gene (RAG)-protein-mediated somatic recombinationv, a process that yields a diverse repertoire of receptors. This diversity is further increased by boosted mechanisms, such as non-templated nucleotide improver, gene conversion and (in the case of B cells) somatic hypermutation, generating a highly various repertoire of receptors with the potential to recognize almost any antigenic determinant in a specific mannerv.
There are 2 types of lymphocyte that express antigen receptors: conventional lymphocytes and innate-like lymphocytes. In the instance of conventional lymphocytes — that is, conventional T cells (near αβ T cells) and B cells (also known as B2 cells) — antigen receptors are assembled essentially at random. Past contrast, for innate-like lymphocytes — that is, B1 cells, marginal-zone B cells, natural-killer T cells and subsets of γδ T cells — the diversity of antigen receptors is restricted and not entirely random. Their specificities are skewed towards a predefined set of ligands37.
The specificities of the receptors of conventional lymphocytes are non predetermined and neither, therefore, is the site where these cells might see their cognate antigen (that is, the antigen specifically recognized by the receptor) or the effector response they need to elicit on activation. So these lymphocytes broadcast through the lymph nodes, which bleed most of the body'due south tissues and organs, and the spleen, which filters the blood, until they encounter an antigen that they are specific for. Microbial antigens are taken up by antigen-presenting cells in the peripheral tissues and are delivered to the lymph nodes or spleen through the lymph or claret, respectively, where they are recognized by conventional lymphocytes. Because the specificity of each antigen receptor is non direct linked to the origin of the antigen, conventional lymphocytes need to be able to differentiate into several types of effector cell, depending on the class of pathogen they recognize (discussed later). The differentiation of conventional lymphocytes into a particular effector-cell type and their localization to the site of infection are regulated past the instructions provided by the innate allowed system, generally in the class of cytokines and chemokines, respectively.
There are 2 types of conventional αβ T cell: T-helper (TH) cells, which are marked by the co-receptor CD4 on the prison cell surface; and cytotoxic T cells, which limited CD8. These cells recognize antigenic peptides bound to major histocompatibility circuitous (MHC) class Ii and class I molecules, respectively. Conventional B cells can recognize well-nigh any antigen by bounden to a specific iii-dimensional molecular determinant (or epitope).
Innate-like lymphocytes differ from conventional lymphocytes in several important ways. Although the antigen receptors of innate-like lymphocytes are assembled in a similar way to those of conventional lymphocytes, their assembly process is not entirely random. Receptor diversity is biased towards a characteristic gear up of specificities for each subset of innate-like lymphocytes37. Accordingly, the effector functions of these lymphocytes and the sites where they reside are often predetermined. The effector responses of innate-similar lymphocytes therefore exercise not more often than not crave the same types of instruction that are provided by the innate immune system to conventional lymphocytes.
The innate-like B cells known every bit B1 cells reside in the peritoneal and pleural cavities and produce mainly antibodies of the IgM form with specificities skewed towards some common bacterial polysaccharides and some cocky antigens38. Innate-like T cells recognize not-classical MHC molecules (too known as MHC class Ib molecules), which can present bacteria-specific ligands: for example, bacterial lipids or formylated peptides in the instance of the CD1 and H–2M3 families, respectively. In a way, these MHC-like molecules function as PRRs, presenting microbial ligands to specialized T cells39. Some not-classical MHC molecules might themselves be ligands for T-cell receptors, without presenting any other molecules. In this example, the expression of these molecules is thought to exist inducible by the engagement of PRRs on specific cell types, such as mucosal epithelial cells40.
Modules of the innate immune system
Different the adaptive allowed organization, the innate allowed organisation is non a single entity. Information technology is a collection of distinct subsystems, or modules, that appeared at dissimilar stages of evolution and behave out different functions in host defence. Some of the master modules found in mammals and how these function in innate host defense force are described in this section (Table 1).
Mucosal epithelia
All metazoans accept mucosal epithelia, one of the most ancient and universal modules of innate immunity. Together with the skin, the mucosal epithelia are the main interface betwixt the host and the microbial world (including both pathogenic and symbiotic microorganisms). Mucosal epithelia have many important functions in protecting the host from pathogen invasion, as well as in establishing a symbiotic relationship with the human being microbiota. Accordingly, mucosal epithelial cells and skin keratinocytes have specialized antimicrobial functions: for case, producing antimicrobial peptides, which limit the viability and multiplication of pathogens and symbiotic microorganisms that colonize these sites. The product of these antimicrobial molecules is induced past appointment of TLRs and NOD proteins and, presumably, other PRRs. Epithelial cells at the mucosal surface likewise produce mucins, which assistance to prevent the zipper and entry of pathogens.
Phagocytes
The phagocytic uptake of pathogens is crucial for host defence force and is carried out by macrophages and neutrophils. These phagocytes are equipped with multiple antimicrobial mechanisms that are activated on initial contact with pathogens. They take a crucial role in defense force confronting both intracellular leaner and extracellular leaner, besides as fungal pathogens. Phagocytosis is facilitated by opsonins, which are host products of the acute-stage response and the complement systems (discussed in the next section), through their ability to bind to both the prison cell walls of microorganisms and the opsonin receptors present on phagocytes.
Acute-stage proteins and complement
A multifariousness of secreted proteins that function in the circulation and tissue fluids — acute-stage proteins and the complement system — constitute another module. Acute-phase proteins are secreted by hepatocytes in response to the pro-inflammatory cytokines IL-1β and IL-6, and the serum concentration of acute-phase proteins increases markedly at the early on stages of infection. A key component of this response is the secreted PRRs: collectins, ficolins and pentraxins41,42,43. Their main functions are opsonizing microbial cells for phagocytosis and activating the complement system. Whereas collectins and ficolins initiate the lectin pathway of complement activation, pentraxins activate the classical pathway, which is besides induced by antibodies41,42,43. Complement activation itself has several consequences, including the following: opsonization of pathogens, through the covalent attachment of C3 fragments; recruitment of phagocytes to the site of infection, through the release of proteolytic fragments of C4 and C5 that take chemotactic activity; and direct killing of pathogens, through the formation of the membrane-attack complex, which is the terminal component of the complement pour44.
Inflammasomes
Inflammasomes are protein complexes that activate pro-inflammatory caspases25. The activation of caspase ane, in detail, is required for processing the IL-1 family of cytokines, including IL-1β, IL-18 and IL-33. These complexes might too process proteins other than pro-inflammatory cytokines. Inflammasomes are activated by the NALP and NAIP subfamilies of NLRs (discussed earlier) in response to bacterial infections and some forms of cellular stress. IL-1-family unit cytokines take diverse functions in inflammation and host defence.
Natural killer cells
Natural killer (NK) cells are specialized in defence against intracellular pathogens, mainly viruses. These cells have 2 main functions: inducing the apoptosis of infected cells and producing cytokines, particularly IFN-γ. They limited two types of receptor, activating and inhibitory, and these receptors recognize their cognate, host-encoded ligands on infected (target) cells45. The residue of expression of activating and inhibitory ligands past a target cell is thought to determine whether it is killed or spared by a particular NK cell. The mechanisms that control the production of these ligands are poorly understood but might involve cell-democratic viral recognition by intracellular sensors of infection or cell-democratic detection of excessive cellular stress. Recognition of viral infection by the infected cells themselves, through RIG-I or MDA5, and by plasmacytoid dendritic cells, through TLRs, likewise controls NK-cell action, by eliciting the production of blazon I IFNs either directly or indirectly through the expression of IL-xv. IL-15 besides regulates NK-cell maintenance46.
Type I IFNs and IFN-induced proteins
Type I IFNs and IFN-induced proteins have a crucial role in defence against viruses. Type I IFNs are produced in response to viral infections, and these proteins trigger the expression of more than 100 genes, the products of which have diverse antiviral activities47. Blazon-I-IFN production can be elicited in ii ways: first, past intracellular sensors of infection (equally described in the previous section); and, second, by TLR3, TLR7 and TLR9 (which are located intracellularly, on endosomes). The start mode of production is ubiquitous and occurs in virally infected cells. It results in autocrine or paracrine IFN-mediated signalling, which confers an antiviral state on the infected cell and neighbouring cells. Past dissimilarity, the second way of production involves the engagement of TLR7 or TLR9, which results in specialized type-I-IFN-producing cells, known as plasmacytoid dendritic cells, producing systemic levels of IFN-α48. In almost all cases, blazon I IFNs are produced in response to viral or bacterial nucleic acids. The only exception to this seems to be IFN-β production in response to TLR4 ligands, which are not nucleic acids.
Eosinophils, basophils and mast cells
Eosinophils, basophils and their products form a host-defence force module involved in protection confronting multicellular parasites, such every bit helminths. Mast cells are also a component of this module, although their function is not restricted to protection against parasites49. Mast cells reside in mucosal and connective tissues, whereas eosinophils and basophils are recruited to the sites of infection from the circulation. During bacterial infection, mast cells can exist activated straight past TLRs49. The mode in which parasites activate mast cells, basophils and eosinophils is largely unknown. Recently, however, one of the main parasite-associated cell-wall components, chitin, was establish to induce eosinophil recruitment50. Interestingly, the main defensive strategy that components of this module use against parasites does not seem to target these pathogens directly (although direct furnishings practice occur). Instead, information technology is the host tissues, particularly the mucosal epithelia, smooth muscles and vasculature, that are the main targets of the allowed response. These tissues are affected by the mediators released by mast cells and basophils in a way that limits the spread of parasites and promotes their expulsion from the host. The function of this module is regulated by several cytokines, including IL-4, IL-5, IL-9 and IL-13 (ref. 51).
Evolution and functional organization of the innate immune system
The diverse modules of the innate immune system evolved at dissimilar stages of phylogeny in response to specific challenges imposed by different classes of pathogen. The appearance of distinct innate host-defence modules during evolution too reflects changes in anatomy and physiology as animals evolved. In addition, a given module is not the aforementioned in different animals and might have been expanded or contracted during evolution in response to specific needs. The limerick of the innate host-defence modules in any given brute species is therefore ane of many possible configurations, presumably the optimal one for affording maximum protection in the specific physiological context. Thus, the innate immune organization, although ancient in origin, is not equivalent in different animal phyla or classes or even between different species of the same course. For example, the number, expression and regulation of the antimicrobial molecules known as defensins varies between mammalian species.
More than marked changes in the structure of the innate immune system tin can be seen at different stages of phylogeny. NK cells, type I IFNs, eosinophils and basophils are all unique to vertebrates. Moreover, the elimination of virally infected cells by NK cells is a viable defensive strategy only in circuitous metazoans, which have renewable tissues, and not in invertebrates, which consist of mail-mitotic cells, near of which exercise not self-renew. (The extreme tissue and organ autonomy and regenerative capacity in plants might similarly explain why immune-response-associated prison cell death is one of the main host-defence strategies in establish amnesty.) In improver, mast-jail cell-mediated and basophil-mediated defence against multicellular parasites is based, in role, on the effects of these cells on the vasculature, which is absent in invertebrates with an open up circulatory organization. Also, most arthropods are not suitable hosts for helminths because they are not large plenty to adjust them. Accordingly, the components of innate immunity confronting parasites are absent-minded from well-nigh or all invertebrate phyla.
Some other of import aspect of the organization of the innate immune system is that modules that are co-induced by an infection tend to develop functional links and are usually co-regulated by the same inducible signals, most normally cytokines. For example, phagocytes, the complement system and astute-stage proteins are functionally linked through opsonization-dependent phagocytosis, and these two modules are co-induced during many bacterial infections. A more specific example is that TLR-activated macrophages produce IL-half dozen, which induces hepatocytes to secrete opsonins during the acute-phase response. Similarly, NK cells and type I IFNs are co-induced by viral infections and are functionally coupled. Not all modules of the innate immune organisation are co-induced by a given infection, withal. The modules that are not functionally coupled (for example, NK cells and basophils) are triggered by singled-out pathways and are not co-regulated by the same cytokines.
The modules of innate host defense are activated by primary sensors of infection, in most cases by PRRs (equally discussed earlier). TLRs tin activate multiple modules (mucosal epithelium, phagocytes, acute-phase proteins and type I IFNs), whereas other PRRs seem to exist more specialized.
Innate control of adaptive immune responses
In addition to direct activation of innate host-defense force mechanisms, some PRRs are coupled to the induction of adaptive immune responses. As discussed earlier, conventional lymphocytes (virtually αβ T cells and B2 cells) limited antigen receptors with random specificities and therefore recognize antigens that lack any intrinsic characteristics indicative of their origin. Therefore, conventional lymphocytes require instructions indicating the origin of the antigen they recognize. These instructions come up from the innate immune system in the form of specialized signals inducible by PRRs4, which tin can sense infection because of their specificity for products of microbial origin. Therefore, the basic principle of innate command of adaptive immunity is based on establishing an clan betwixt the antigens recognized by lymphocytes and the microbial products (that is, PAMPs) recognized past PRRs.
For T cells, this association is interpreted by dendritic cells. Dendritic cells reside in most peripheral tissues, where they monitor the tissue surroundings for the presence of pathogens past using various PRRs. When a pathogen is encountered past a dendritic cell, it is taken up past phagocytosis, and its protein constituents are candy into antigenic peptides, which are presented at the jail cell surface by MHC class I and/or grade II molecules. For MHC form Ii molecules, the antigenic peptides selected for presentation derive from the phagosome in which the pathogen was internalized in response to the triggering of TLRs or other PRRs52. A like mechanism might also operate for MHC grade I molecules. Therefore, the association between an antigen and a PAMP is established as a result of their presence in the same phagocytosed 'cargo' (for example, a bacterial cell). PRRs likewise activate dendritic cells, inducing them to produce cytokines and limited jail cell-surface signals and to drift to the lymph nodes through the lymphatic vessels that drain the site of infection. When these dendritic cells reach the lymph nodes, they nowadays the pathogen-derived antigens, together with PRR-induced signals (cytokines and cell-surface-associated molecules), to T cells. This results in T-cell activation and, in the example of TH (CD4+) cells, differentiation into one of several types of effector TH cell53.
For B cells, the clan between an antigen and a PAMP tin can be established directly, when the two are physically linked in a single molecule or particle. This presumably occurs through co-engagement of a B-cell receptor and a PRR. In the extreme case, a TLR ligand (for example, lipopolysaccharide or flagellin) is itself recognized past the B-cell receptor and by a corresponding TLR expressed past a B jail cell. Antigens of this class, which combine ligands for both innate and adaptive allowed recognition, are called T-contained antigens, because they can elicit B-cell responses without 'help' from TH cells. When an antigen and a PAMP are non physically linked, their association is established through effector TH cells that take previously been activated by dendritic cells. Antigens of this grade (commonly proteins) are called T-dependent antigens.
The antigen receptors of innate-like lymphocytes are skewed towards the recognition of microbial products, so the activation of these cells does not crave the same elaborate mechanisms every bit for conventional lymphocytes. Indeed, B1 cells can be activated directly by PRRs and are programmed to produce antibodies with a wide specificity for common bacterial antigens38. Innate-like T cells recognize microbial antigens (such as lipids, glycolipids and formylpeptides) presented by non-classical MHC molecules. In certain cases, these cells recognize MHC-like molecules that practise not seem to present whatsoever antigens but whose expression is inducible by PRRs. In such cases, the product of T-cell-receptor ligands in response to microbial products might be sufficient to signal the presence of infection.
TH cells tin can differentiate into several types of effector cell: TH1, THtwo and TH17 cells54 (Fig. 1). These cells are characterized by the product of singled-out sets of cytokines55,56. THone cells produce IFN-γ and activate macrophages and other cell types to trigger defence force confronting intracellular pathogens. TH1-cell-derived IFN-γ also instructs B cells to produce antibodies of the IgG2 subclass. THtwo cells are involved in protection against multicellular parasites and produce IL-4, IL-5 and IL-xiii (ref. 51). These cytokines control the role of eosinophils, basophils and the mucosal epithelia. IL-4 also instructs B cells to produce antibodies of the IgE class, which are important in defense force against parasites through their effects on mast-cell and basophil activation57. Finally, TH17 cells produce IL-17, which induces not-haematopoietic cell types, including epithelial cells, to produce chemokines that recruit neutrophils to the site of infection58. TH17-prison cell responses are involved in protection against extracellular bacteria and fungi14. The differentiation of naive TH cells (which take non previously encountered their cognate antigen) into the three effector-prison cell lineages, TH1, TH2 and TH17 cells, is controlled by transcriptional master regulators, in this example T-bet, GATA-bounden protein three (GATA3) and retinoic-acid-receptor-related orphan receptor-γt (RORγt), respectively59. The expression of these master regulators is controlled by cytokines produced by antigen-presenting cells (such equally dendritic cells) in response to PRR activation.

When circulating 'naive' TH cells first recognize their cognate antigen, they differentiate into one of several effector-jail cell lineages (listed in assuming), depending on the infecting pathogen. TH1, TH2 and TH17 cells are the known types of effector TH cell; however, other types of effector TH prison cell probably exist. Each TH-cell lineage is characterized past the cytokines that are produced and by the innate immune effector mechanism that is activated (denoted by arrows). Information technology is possible (but has not been proved) that every module of the innate immune organization is controlled past a dedicated effector TH-prison cell lineage.
The effector response in each example is thus dictated past the innate immune arrangement. In terms of TH cells, TLR appointment induces IL-12 product, which directs TH cells to differentiate into TH1 cells. By contrast, TLR-induced IL-vi, together with transforming growth factor-β (from an unknown cellular source), induces differentiation into TH17 cells58,59. And dectin-1 date results in the production of IL-23, which is required for TH17-cell function and/or maintenance17,60. The mechanisms of THii-cell generation are unknown but presumably follow a similar principle, with the defended cytokines probable to be IL-4 and thymic stromal lymphopoietin (TSLP), produced in response to engagement of an unidentified sensor subsequently helminth infection61. For other cell types, blazon I IFNs (which are produced in response to TLR date or RIG-I, MDA5 or DAI appointment during viral infections) regulate the office of cytotoxic T cells and NK cells, either directly or indirectly past inducing IL-15 production62.
Chiefly, the adaptive immune response ultimately results in an antigen-specific activation of the effector mechanisms of the innate immune system. Thus, the effector TH cells produce the appropriate effector cytokines that actuate a specific module of the innate allowed organization (Fig. 2), including activation of macrophages by TH1 cells, activation of neutrophils past TH17 cells and activation of eosinophils, mast cells and basophils past THii cells54,63. Similarly to NK cells, cytotoxic T cells induce apoptosis of infected cells, except that the T-cell response is antigen specific. Too, antibodies activate the modules of the innate immune system in a class-dependent (and antigen-dependent) way. IgG activates complement and opsonizes pathogens to aid their phagocytosis by macrophages and neutrophils, whereas IgE activates mast cells and basophils. Each of the innate effector responses can therefore be activated either directly, by the appropriate PRRs at the early on stages of infection, or indirectly, by T cells and antibodies (in an antigen-specific manner) at the subsequently, effector, stages of the immune response (Fig. two). Furthermore, each effector machinery of the adaptive allowed organization might have evolved to activate the appropriate host-defence module of the innate allowed arrangement.
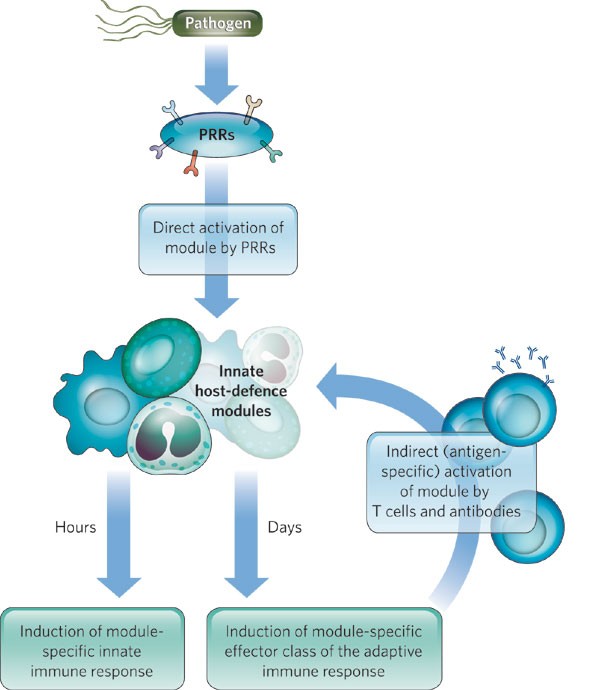
Host-defence mechanisms tin can be induced straight, by engagement of PRRs, or indirectly, by T cells and/or antibodies. Each module is characterized by singled-out antimicrobial defence mechanisms and can instruct the adaptive immune system to mountain a response involving a module-specific effector class. After an adaptive immune response has been initiated, it results in antigen-specific activation of the same innate immune module that instructed the adaptive immune response. For instance, macrophages can exist activated either directly by TLRs or indirectly by TH1 cells, through IFN-γ, CD40 ligand and other signals. Eosinophils tin exist activated either direct by an unidentified PRR or indirectly by TH2 cells. And the classical pathway of complement activation can be induced either directly by pentraxins or indirectly past antibodies. Antigen-specific activation of the innate host-defence modules is more efficient than direct activation and is oft required for pathogen clearance.
The relative contributions of the innate immune organization and the adaptive immune organisation during bacterial infections accept been investigated extensively. 1 of import principle that has emerged from these studies is that, although innate host defence is crucial for controlling an infection, it is frequently insufficient for pathogen clearance64. For case, to clear a Listeria monocytogenes infection requires functional T-prison cell responses64. It therefore seems that the innate immune system in vertebrates evolved to depend, to some extent, on antigen-specific (adaptive) immunity. This might explain why the mammalian innate allowed organization, unlike that of arthropods, is not cocky-sufficient at affording protection against many infections. It should be noted, still, that our understanding of host defence might be biased because almost all studies are based on symptomatic infections. Asymptomatic infections are presumably mutual, and many of these infections might be cleared efficiently past innate host-defence force mechanisms.
Conclusions and perspectives
The adaptation of microorganisms to host niches tin benefit the host, equally is the example with many symbiotic microorganisms. In some cases, withal, adaptation negatively affects tissue physiology and might direct damage tissues, resulting in symptomatic communicable diseases. The symptoms of an infectious disease can also exist caused past excessive immune and inflammatory responses, and these can often be more dissentious to the host than the virulence activity of the pathogen that elicited them. Thus, it is just every bit crucial for the host to limit the immunopathology equally it is to protect against the infecting pathogen. The trade-off betwixt immunopathology and protection against infection has presumably resulted in an optimal balance of the sensitivity and intensity of the immune response. This rest is probably not difficult-wired, because the immune response needs to vary in intensity and elapsing depending on the infecting pathogen. One possible solution to the conundrum of how the balance is achieved would be that the tissue damage acquired directly by the pathogen tin exist distinguished from the damage inflicted by the immune response. If this is the example and the two types of tissue damage are differentially detected past the host, then the extent of the impairment might negatively control the intensity and duration of the immune response. Understanding the remainder between the ii alien causes of infectious-illness symptoms is crucial for the evolution of appropriate therapeutic strategies65.
Another surface area of great importance is agreement the principles of protective amnesty. What matters to the host organism is not the induction of an immune response but whether the immune response protects against a given infection. Not all immune responses are protective. Only immune responses of the correct effector class directed at particular antigens tin can provide protection against infection. Information technology is generally assumed that the effector response depends on the pathogen type. Yet, in nearly cases, the crucial pathogen features that determine the effector immune response are unclear. Similarly, information technology is not known how the relevant features of pathogens are translated into the appropriate fix of signals that make up one's mind the effector response. Understanding these principles is vital for the development of vaccines that can elicit protective immunity.
Source: https://www.nature.com/articles/nature06246
0 Response to "2a Review How Does the Immune System Identify a Pathogen?"
Post a Comment